The Great Debate: Is Cannabis a Supplement?

The Confusing World of Cannabis Classification
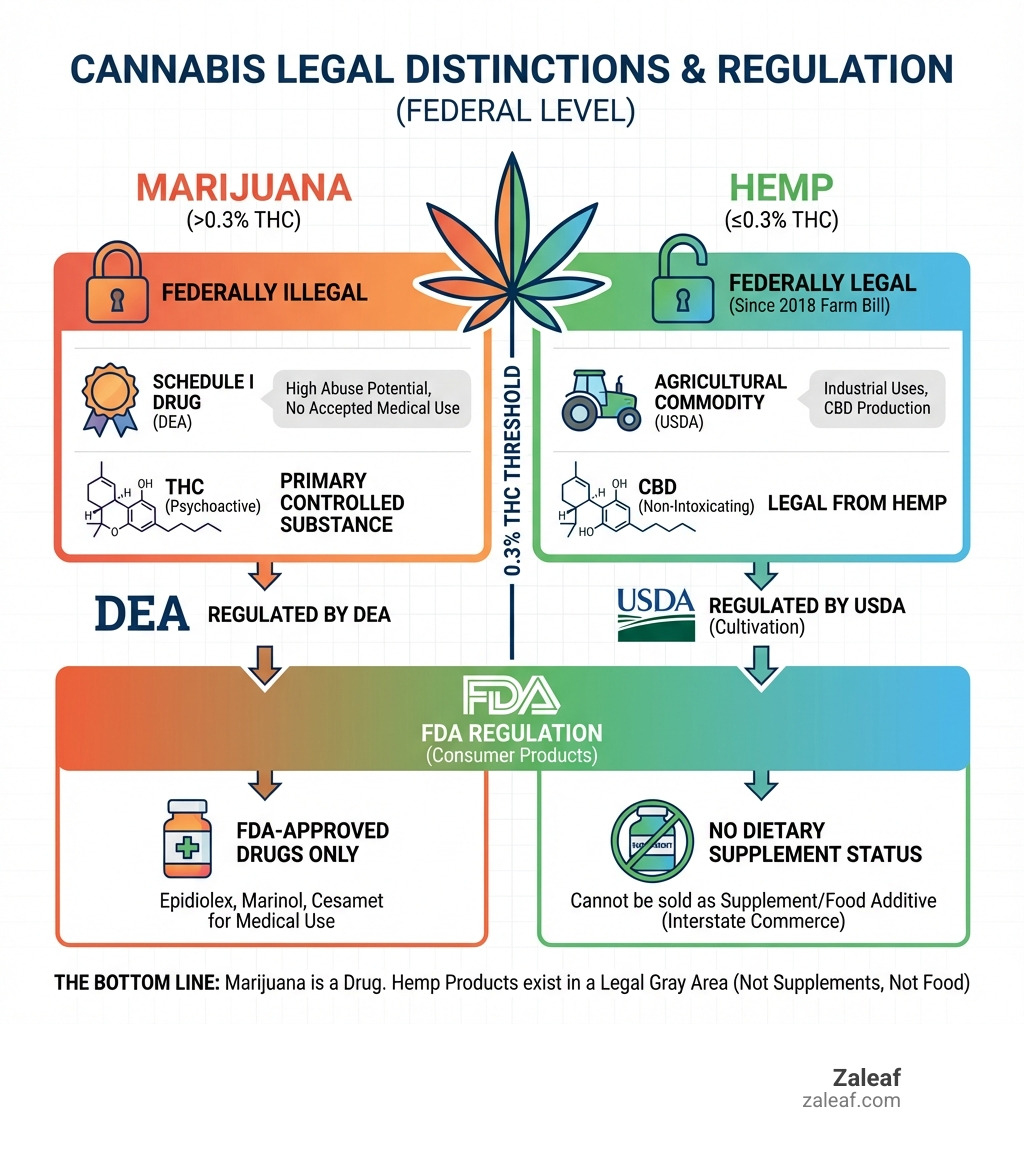
Is cannabis a drug or supplement? The answer depends on where you are, what type of cannabis you're using, and who's regulating it. Here's what you need to know:
Federal Classification:
- Marijuana (cannabis with more than 0.3% THC) is classified as a Schedule I controlled substance — legally defined as a drug with high abuse potential and no accepted medical use
- Hemp (cannabis with 0.3% THC or less) was removed from the Controlled Substances Act in 2018 and is legal federally
Regulatory Status:
- The FDA does not allow cannabis products (THC or CBD) to be sold as dietary supplements
- Cannabis cannot legally be added to food in interstate commerce
- Only FDA-approved cannabis-derived drugs (like Epidiolex, Marinol, and Cesamet) can be marketed for medical use
The Bottom Line: Under federal law, marijuana is a drug. Hemp-derived products exist in a legal gray area — they're not supplements, not food additives, but they're legal to sell if they meet specific criteria.
This classification conflict creates real confusion for consumers seeking legal, tested cannabis products for stress relief and relaxation. You might see CBD oils marketed online, cannabis edibles in dispensaries, and hemp products in wellness stores — all treated differently under the law.
As Max Shemesh, Owner & CEO of Zaleaf, I've spent years navigating this regulatory maze to bring high-quality cannabinoid products to customers who need clarity on is cannabis a drug or supplement. Through building partnerships with over 300 retailers and staying ahead of evolving regulations, I've learned that understanding these distinctions is essential for anyone seeking safe, legal cannabis products.
Is cannabis a drug or supplement helpful reading:
The Federal Stance: Why Cannabis is a Schedule I Drug
When we talk about is cannabis a drug or supplement, the federal government's perspective is the first and most critical piece of the puzzle. According to federal law, specifically the Controlled Substances Act (CSA) enacted in 1970, cannabis (or marijuana, as it's often referred to in legal texts) is unequivocally classified as a Schedule I controlled substance. This classification is the bedrock of federal cannabis prohibition and shapes virtually every aspect of its legal and commercial status in the United States.

What exactly does "Schedule I" mean? It's a designation reserved for drugs that the federal government believes have:
- A high potential for abuse.
- No currently accepted medical use in treatment in the United States.
- A lack of accepted safety for use under medical supervision.
This definition, established over 50 years ago, means that from a federal standpoint, the cultivation, manufacture, possession, and distribution of marijuana are illegal, except for sanctioned research. This sweeping prohibition applies regardless of whether a state has legalized cannabis for medical or recreational purposes. It's a key reason why the debate over is cannabis a drug or supplement is so complex.
The history of federal prohibition dates back to the early 20th century, with significant legislative milestones like the Marihuana Tax Act of 1937 and the 1970 CSA solidifying its status as an illicit substance. This historical context is vital for understanding the current regulatory landscape.
The Role of the DEA and DOJ
The Drug Enforcement Administration (DEA) is the federal agency tasked with enforcing the controlled substances laws and regulations of the United States. Their mission is to "bring to the criminal and civil justice system of the United States... those organizations and principal members of organizations, involved in the growing, manufacturing, or distribution of controlled substances appearing in or destined for illicit traffic in the United States." Given cannabis's Schedule I status, it falls squarely within the DEA's enforcement purview.
The Department of Justice (DOJ) has historically echoed support for this position, maintaining that cannabis is a significantly harmful substance with no recognized medical uses at the federal level. While federal enforcement priorities have shifted over time, largely focusing on major traffickers rather than individuals complying with state laws, the underlying federal prohibition remains. This creates a challenging environment for businesses and individuals operating in state-legal cannabis markets, as they technically remain in violation of federal law. For more detailed information on the criteria for drug scheduling, you can refer to the DEA's drug scheduling criteria.
The Impact of the 2018 Farm Bill
A significant development in the federal stance on cannabis, albeit a nuanced one, came with the 2018 Farm Bill. This landmark legislation introduced a crucial distinction between "marijuana" and "hemp." Under the 2018 Farm Bill, hemp is defined as Cannabis sativa L. plant material containing no more than 0.3% delta-9-tetrahydrocannabinol (THC) on a dry weight basis. This seemingly small percentage difference had monumental implications: hemp was removed from the Controlled Substances Act and is no longer a federally controlled substance.
This meant that hemp and its derivatives, including CBD products derived from hemp, became federally legal, provided they meet the 0.3% THC threshold. This legislative change paved the way for the burgeoning CBD market and the rise of other hemp-derived cannabinoids. The U.S. Department of Agriculture (USDA) now plays a role in regulating hemp cultivation, overseeing state and tribal hemp programs.
However, understand that this change did not legalize marijuana. Cannabis plants exceeding the 0.3% delta-9 THC limit remain Schedule I controlled substances. This distinction is paramount to understanding why some cannabis products are legal while others are not, and why the question of is cannabis a drug or supplement remains contentious. We dive deeper into this topic in our guide, Decoding the Farm Bill: An Explainer on Hemp and CBD Legality.
So, Is Cannabis a Drug or Supplement? The Regulatory Maze
Navigating the regulatory landscape of cannabis feels like being lost in a funhouse mirror maze – everything looks familiar, but nothing is quite as it seems. The fundamental question, is cannabis a drug or supplement, becomes even more convoluted when we consider the various regulatory bodies and their overlapping, yet often conflicting, definitions.
The U.S. Food and Drug Administration (FDA) is the primary federal agency responsible for regulating food, drugs, cosmetics, and dietary supplements under the Food, Drug, and Cosmetic Act (FD&C Act). Their authority is broad, and their stance on cannabis products is clear: they treat cannabis and cannabis derivatives as drugs that must be approved by the FDA before being marketed to consumers. This creates an inherent tension with state laws that have allowed cannabis products to be sold in various forms.
Businesses operating in the cannabis space often eye three primary avenues for marketing their products: as food, as dietary supplements, or as drugs. However, each path is fraught with federal regulatory problems.
Why can't cannabis products be sold as dietary supplements?
This is where the debate over is cannabis a drug or supplement gets particularly tricky for many consumers. The Dietary Supplement Health and Education Act of 1994 (DSHEA) provides a framework for dietary supplements, which are meant to supplement the diet and contain one or more dietary ingredients (like vitamins, minerals, herbs, or amino acids). However, the FDA has explicitly concluded that products containing THC and CBD are generally excluded from the dietary supplement definition.
Why? Because both THC and CBD have been studied as active ingredients in FDA-approved or extensively investigated drugs. THC is the active ingredient in Marinol and Syndros (approved in 1985 and 2016, respectively), and CBD is the active ingredient in Epidiolex (approved in 2018). Under federal law, if an active ingredient has been authorized for investigation as a new drug, or if it's the active ingredient in an approved drug, it cannot typically be marketed as a dietary supplement or food, unless it was marketed as such before the drug investigations or approval. Since the clinical investigations for THC and CBD occurred before widespread marketing as supplements, the FDA maintains this stance.
The FDA has not shied away from enforcement. They've issued numerous warning letters to companies illegally selling CBD products, especially those making unproven therapeutic claims or marketing CBD as a dietary supplement. This firm regulatory position by the FDA significantly impacts the ability to categorize cannabis as a supplement. For more on the FDA's regulatory approach, see FDA regulation of cannabis products.
What About Edibles and Foods?
If not a dietary supplement, then can cannabis be considered food? Many states, like Illinois, allow for cannabis-based edibles such as cookies or brownies. Some state legislation even refers to these products as "food." However, the FDA’s position is clear: it is prohibited to add THC or CBD to food (including animal food) in interstate commerce.
Under federal law, adding a substance that is an active ingredient in an approved drug or a drug under substantial clinical investigation to food would render that food "adulterated" under the FD&C Act. This means that federally, cannabis-infused edibles are not recognized as legitimate food products. In fact, some state laws, despite legalizing edibles, still require them to be labeled with disclaimers like "not a food" to avoid FDA food-labeling requirements. This dichotomy further illustrates the federal-state conflict that complicates the simple question: is cannabis a drug or supplement? For a deeper dive into this, check out our article on Cannabis Edibles Explained: Are They Safe and Effective?.
The Path to an FDA-Approved Drug
Given the federal government's stance, the most legitimate pathway for cannabis and its derivatives to be recognized for medical use is through the rigorous FDA drug approval process. This involves:
- Investigational New Drug (IND) Application: Companies must submit an IND application to the FDA to conduct clinical trials on a cannabis-derived compound. This is a crucial step to demonstrate safety and efficacy. You can learn more about this process here: Investigational New Drug (IND) application.
- Clinical Trials: Multiple phases of human clinical trials are required to gather data on the drug's safety, dosage, and effectiveness for specific conditions. This is a time-consuming and incredibly expensive endeavor.
- New Drug Application (NDA): If trials are successful, a company submits an NDA, presenting all the scientific data for FDA review.
Currently, the FDA has approved a few cannabis-related medicines:
- Epidiolex: A purified liquid form of cannabidiol (CBD), approved for treating seizures associated with Lennox-Gastaut syndrome, Dravet syndrome, and tuberous sclerosis complex in patients 1 year of age and older.
- Dronabinol (Marinol, Syndros): Synthetic THC, approved for treating nausea and vomiting caused by cancer chemotherapy and appetite loss in AIDS patients.
- Nabilone (Cesamet): A synthetic cannabinoid chemically similar to THC, also approved for chemotherapy-induced nausea and vomiting.
These approvals highlight that the FDA does recognize the therapeutic potential of specific, isolated cannabis compounds when they undergo the stringent drug development and approval process. However, the cannabis plant itself, or broad-spectrum extracts, have not received FDA approval as medicine. This demanding process, with its high costs and extensive time commitment, is why so few cannabis-derived drugs have made it to market.
The State-Level Patchwork and the Rise of Wellness Cannabis
While the federal government maintains its strict classification of marijuana, the states have been telling a very different story. This creates a complex and often contradictory "federal-state policy gap" that profoundly impacts the question of is cannabis a drug or supplement.
Since California became the first state to allow for medical marijuana in 1996, a wave of state-level reforms has swept across the nation. As of April 1, 2022:
- 37 states, plus the District of Columbia, Puerto Rico, Guam, and the U.S. Virgin Islands, have comprehensive laws allowing for the medical use of marijuana.
- An additional 11 states allow for the medical use of low-THC cannabis.
- 18 states, the District of Columbia, Guam, and the Northern Mariana Islands allow for the recreational use of marijuana.
This means that a significant majority of Americans now live in areas where cannabis is legal in some form, at least under state law. This rapid evolution has led to a thriving state-legal cannabis industry, despite the lingering federal prohibition.
Medical vs. Recreational Use: What's the Difference?
The distinction between medical and recreational cannabis laws often comes down to accessibility, qualifying conditions, and possession limits.
- Medical Marijuana Laws: These typically require patients to obtain a recommendation from a doctor for a specific "qualifying condition" (e.g., chronic pain, epilepsy, cancer, MS). Patients then receive a medical card, allowing them to purchase cannabis from licensed dispensaries, often with higher possession limits and lower taxes than recreational products.
- Recreational Marijuana Laws: These allow adults (usually 21 and older) to purchase and possess specified amounts of cannabis without a medical recommendation. While they offer broader access, recreational markets often have stricter possession limits and higher taxes.
The existence of medical marijuana programs directly challenges the federal Schedule I assertion that cannabis has "no currently accepted medical use." This state-level recognition of cannabis's therapeutic potential is a key factor in the public debate over is cannabis a drug or supplement. For those wondering if a medical card is still needed, we explore this in Is No Medical Card Needed? The Future: A Legal Perspective.
Navigating the legal landscape: Is cannabis a drug or supplement today?
The 2018 Farm Bill's legalization of hemp created a new category of cannabis products: hemp-derived cannabinoids. Since hemp is no longer a Schedule I controlled substance, cannabinoids extracted from hemp, such as CBD, CBG, CBN, and even certain forms of THC like Delta-8 THC, exist in a complex legal space.
These hemp-derived products are often marketed in ways that blur the lines between traditional supplements and regulated drugs. While the FDA maintains that CBD and THC cannot be sold as dietary supplements, the federal legality of hemp itself has allowed a massive market for these products to flourish. This is where companies like Zaleaf focus our efforts, ensuring that our products are 100% legal, derived from hemp, and compliant with the 0.3% Delta-9 THC federal limit.
The rise of cannabinoids like Delta-8 THC further complicates the picture. Delta-8 THC is a psychoactive cannabinoid found in the cannabis plant that can be derived from legal hemp. Its legal status is often debated, but many states permit its sale under the umbrella of hemp legality. Understanding these nuances is crucial for consumers. We have several articles that dig into this, including The Legal Landscape of Delta-8 THC Products.
In this environment, the importance of product sourcing and third-party lab testing cannot be overstated. With a lack of comprehensive federal regulation for hemp-derived products, it falls to responsible companies to ensure product safety, potency, and purity. Our commitment at Zaleaf is to provide transparency and peace of mind through rigorous third-party testing, so our customers always know exactly what they're getting.
Weighing the Evidence: Potential Benefits vs. Known Risks
When we consider is cannabis a drug or supplement, look at the scientific evidence regarding its effects on the human body. Research into cannabis, despite federal restrictions, has illuminated both promising therapeutic potential and important health risks.
| Potential Therapeutic Uses (Drug-like) | Known Health Risks & Side Effects (Drug-like) |
|---|---|
| Chronic pain (neuropathic, cancer-related) | Cognitive impairment (memory, attention, judgment) |
| Nausea and vomiting (chemotherapy-induced) | Mental health risks (anxiety, psychosis, depression) |
| Epilepsy (Lennox-Gastaut, Dravet, TSC) | Cardiovascular effects (increased heart rate, blood pressure) |
| Multiple Sclerosis (MS) spasticity | Cannabis Use Disorder (addiction, withdrawal) |
| Appetite stimulation (AIDS-related wasting) | Lung health issues (bronchitis, inflammation from smoking) |
| Sleep problems | Gastrointestinal problems (Cannabinoid Hyperemesis Syndrome) |
| Anxiety and stress relief | Drug interactions (alcohol, anticoagulants, CNS depressants) |
The cannabis plant contains over 100 active compounds called cannabinoids, with Delta-9 Tetrahydrocannabinol (THC) and Cannabidiol (CBD) being the most well-known. These compounds interact with our body's endocannabinoid system (ECS), a complex network involved in regulating various physiological processes, including mood, pain, appetite, and memory. This interaction is key to both the plant's potential benefits and its side effects.
Researched Health Benefits and Uses
While the FDA has only approved a handful of cannabis-derived drugs, a significant body of research and anecdotal evidence points to several potential health benefits of cannabis. This is often why people consider the question is cannabis a drug or supplement from a wellness perspective.
- Chronic Pain: Cannabis, particularly THC, has shown promise in managing chronic pain, including neuropathic pain associated with conditions like HIV and diabetes. It's often used as an alternative or adjunct to opioid pain medications.
- Nausea from Chemotherapy: Dronabinol (synthetic THC) and nabilone (synthetic cannabinoid) are FDA-approved to treat severe nausea and vomiting in cancer patients undergoing chemotherapy.
- Epilepsy (Seizures): Epidiolex, a CBD-based medication, is FDA-approved for specific severe forms of childhood epilepsy, demonstrating CBD's anticonvulsant properties.
- Spasticity (MS): Cannabis can help reduce muscle spasms and stiffness associated with multiple sclerosis (MS), improving quality of life for some patients.
- Anxiety and Stress: Many users report that cannabis, especially certain strains or CBD-dominant products, can help alleviate anxiety and stress, promoting relaxation. We explore this further in Cannabis for wellness: exploring its role in relaxation, pain and stress.
The National Institutes of Health (NIH) and various research institutions are continuously investigating the medical efficacy of cannabis for a wide range of conditions. You can explore current clinical trials on cannabinoids to see the ongoing scientific efforts.
Understanding the Risks and Side Effects
Despite the potential benefits, it's crucial to acknowledge the known health risks and side effects associated with cannabis use, which is why the federal government classifies it as a drug.
- Cognitive Effects: THC can impair short-term memory, attention, and decision-making, especially in adolescents whose brains are still developing. Long-term heavy use, particularly starting at a young age, may be associated with persistent cognitive deficits.
- Mental Health Risks: Cannabis use, particularly high-potency THC, has been linked to an increased risk of developing or exacerbating certain mental health conditions, including anxiety, depression, and psychosis (especially in individuals predisposed to such conditions).
- Cardiovascular Effects: Immediately after use, cannabis can increase heart rate and blood pressure. Some research suggests a potential link between long-term cannabis use and an increased risk of stroke, heart attack, and arrhythmias, though more research is needed to confirm direct causation.
- Cannabis Use Disorder: Regular and heavy cannabis use can lead to dependence and addiction, characterized by a compulsive need for the drug and withdrawal symptoms upon cessation. The percentage of the population 12 and older that reported using marijuana in the past month increased from 6.9% in 2010 to 11.8% in 2020, highlighting the growing prevalence.
- Lung Health (Smoking): Inhaling cannabis smoke, like tobacco smoke, introduces toxins, irritants, and carcinogens into the lungs. This can lead to respiratory issues such as chronic bronchitis, increased airway resistance, and lung inflammation.
- Gastrointestinal Problems: Long-term, heavy cannabis use can lead to Cannabinoid Hyperemesis Syndrome (CHS), characterized by severe, recurrent bouts of nausea, vomiting, and abdominal pain.
- Drug Interactions: Cannabis can interact with various medications, potentially altering their effectiveness or increasing side effects. For example, it can amplify the effects of central nervous system depressants (like alcohol or sedatives), increase the risk of bleeding with anticoagulants, and interfere with certain protease inhibitors and anti-seizure medications. Always consult a healthcare professional about potential interactions, especially if you're on other medications. For more information on potential interactions, consult reputable medical resources or your healthcare provider.
Understanding these risks is vital for informed and responsible use, regardless of whether one views is cannabis a drug or supplement primarily through a medical or wellness lens.
Conclusion: A Path Forward in a Divided Landscape
The journey to understand is cannabis a drug or supplement reveals a landscape marked by profound regulatory divides and evolving public perception. Federally, marijuana remains a Schedule I controlled substance, firmly categorized as a drug with no accepted medical use and a high potential for abuse. This stance, enforced by the DEA and FDA, dictates that cannabis cannot be marketed as a food or dietary supplement and must undergo rigorous drug approval processes to be recognized for medical use.
However, the reality on the ground is starkly different. A growing number of states have legalized cannabis for medical and/or recreational purposes, creating a thriving industry that often treats cannabis products as wellness aids, foods, or therapeutic compounds. The 2018 Farm Bill further complicated matters by legalizing hemp and its derivatives, including CBD and other cannabinoids like Delta-8 THC, provided they contain less than 0.3% Delta-9 THC. These hemp-derived products occupy a unique, often ambiguous, space, allowing for their widespread availability outside traditional drug channels.
So, where does this leave us? The answer to is cannabis a drug or supplement is not a simple either/or.
- As a "drug," it refers to the federally prohibited marijuana plant and specific FDA-approved pharmaceutical compounds derived from cannabis.
- As a "supplement" (in the broad sense of wellness support), it encompasses the vast array of hemp-derived products, like those offered by Zaleaf, that are legally available and used for relaxation, pain management, and stress relief. These products, while not federally recognized as dietary supplements by the FDA, are acceptd by consumers seeking natural alternatives for wellness.
This complex situation underscores the critical need for transparency, quality, and consumer education. At Zaleaf, we are committed to navigating this intricate legal framework to provide 100% legal cannabis products. We understand that our customers rely on us for safe, effective, and compliant options. That's why we prioritize enhancing cannabinoid and terpene profiles for mood-specific effects and ensure all our products undergo rigorous third-party lab testing. We believe that clarity and quality are paramount in this evolving industry.
As the legal landscape continues to shift, and as research further illuminates the potential of the cannabis plant, we remain dedicated to helping our customers explore cannabis for wellness: exploring its role in relaxation, pain and stress. Our goal is to empower you with knowledge and access to high-quality products that meet your needs, all within the bounds of current regulations. The debate may rage on, but our focus remains on delivering trusted, tested, and transparent cannabis solutions.


